Chalkeyes presents: Gene warfare for AMD?
Patients suffering from age-related macular degeneration (AMD) can feel encouraged by the latest therapeutic discoveries.
About AMD
AMD is a progressive condition that damages the retinal pigment epithelium (RPE) and photoreceptors in the retina and is classified as early, intermediate or late-stage AMD. Atrophic or dry AMD is the most common type (85-90% of all cases), is slower progressing and involves age-related ‘wear and tear’ to the macula. Neovascular or exudative AMD, better known as wet AMD, accounts for the remaining 10-15% of AMD patients and is caused by the growth of abnormal blood vessels underneath the macula.
The development of anti–vascular endothelial growth factor (anti-VEGF) agents was a breakthrough in the treatment of wet AMD, however effective dry AMD therapy remains an unmet need.
Prevalence studies confirm several preventable environmental risk factors for susceptibility to AMD, including smoking, hypertension, age itself and body mass index. Soft, indistinct drusen and pigment abnormalities at the macular have a high association with progression to late-stage AMD and research into the constituents of drusen has revealed a genetic component.
AMD and gene therapy
Pioneering research over a decade ago discovered a link between AMD and the activation of the complement cascade, a series of proteins in the blood that helps our immune system cells eliminate invading pathogens.
The complement proteins (CFH, CFB/C2, C3, CF1) and activators found in drusen have established genetic links (Geerlings) and are indicative of early AMD. Studies have shown the AMD risk with CFH polymorphism is approximately 50% and when combined with the CFB/C2 haplotypes (genetic determinants from a single chromosome), AMD can be predicted in 74% of cases (Troubeck).
Since this first vital discovery of CFH presence in AMD in 2005, there have been more than 20 additional genetic risk loci linked to AMD susceptibility. Complement activation also induces angiogenic growth factor release and choroidal neovascularization – a discovery that led to anti-VEGF therapies. Thus, a potential long-term alternative treatment strategy to anti-VEGF agents is gene therapy.
But the complexity of gene therapy is an issue, explained Dr Szilard Kiss, assistant professor of ophthalmology and director of clinical research at Weill Cornell Medical College in New York. “We need to consider if we’re delivering anti-VEGF aflibercept or anti-VEGF ranibizumab or a gene therapy to treat a disorder? How are you delivering that in terms of formulation? Where in the retina - either intravitreal or subretinal space - are you delivering your gene therapy vector?”
Basically, the success of gene therapy for AMD is dependent on the accurate selection of the disease-causing protein and its level of expression in the disease.
Luxturna, was the first gene therapy to be approved for an inherited eye disease, Leber’s congenital amaurosis (LCA). An expensive, one-time treatment for LCA that uses a harmless virus to deliver healthy copies of the gene to the retina, the treatment has had impressive results. This success has prompted a plethora of potential gene therapy studies for other eye diseases, including AMD.
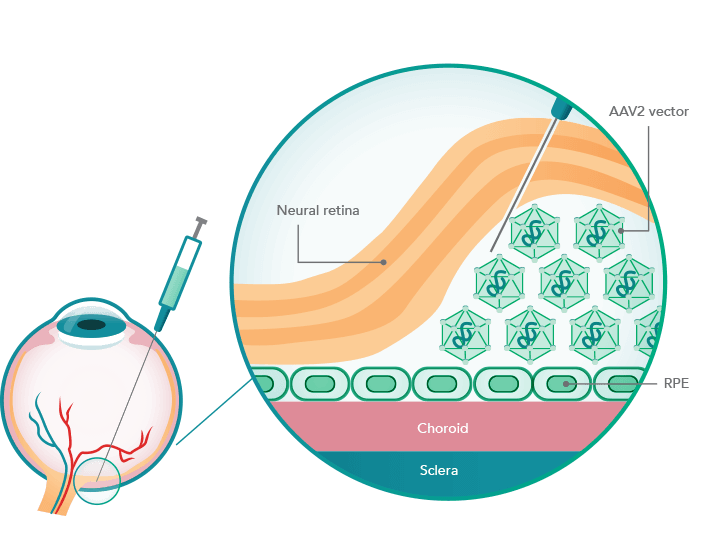
LUXTURNA uses the adeno-associated viral vector serotype 2 (AAV2) to carry a functional copy of the RPE65 gene into the retinal pigment epithelial (RPE) cells to compensate for the RPE65 mutation
Genes can be placed in the eye by an intravitreal injection or injected directly under the retina using a safe virus vector that does not cause infection or harm to the retina. Subretinal delivery allows for broader coverage and a higher protein expression, said Dr Kiss. “There is up to 1,000-fold less expression when you give gene therapy via an intravitreal injection. That’s been shown in non-human primates.”
Neovascular AMD gene therapy
In 2015, Rakoczy et al initiated a subretinal gene therapy study for neovascular AMD targeting the upregulation of VEGF-inhibitor production in the photoreceptor and retinal pigment epithelial cells. Upregulation is the process a cell uses to increase a component, such as RNA or protein, in response to an external stimulus. In this case, the inhibitor was enclosed in a viral vector and inserted into the subretinal space. The researchers found subretinal injection of rAAV.sFLT-1 was highly reproducible, safe and well tolerated.
Then in 2016, Constable et al found that five out of the six subjects treated with sFLT-1 exhibited visual improvement, indicating that sustained delivery of sFlt-1 using subretinal injection may improve patient visual outcomes when compared to intravitreal anti-VEGF administration.
“Since our initial report in 2015, the adeno-associated virus mediated delivery system has emerged as the number one candidate for medical gene therapy,” said Professor Elizabeth Rakoczy, a molecular ophthalmologist at the University of Western Australia when I contacted her for comment. “At the moment a clinical trial conducted by Adverum Biotechnologies is using a modified version of rAAV.sFLT-1 treatment and we anticipate that it might be available for clinical use within three years.”
Adverum was successful in forming a viral vector (AAC.7m8) encoded with aflibercept, administered intravitreally that can transect the retina. The results of a second trial by RegenXBio, investigating AAV8-mediated subretinal delivery of an anti-VEGF antibody fragment (RGX-314), should be published in 2020. Dr Jeffrey Heier, the primary investigating ophthalmologist in this trial and the director of retina research at Ophthalmic Consultants of Boston, presented a trial update in August 2018 that indicated promising results. “50% of patients remained free of anti-VEGF injections after six months... RGX-314 has the potential to provide optimal long-term visual outcomes with a single treatment,” he said.
Extending the treatment regime for intravitreal injections in neovascular AMD is another important focus for research.
New Zealand and Australia participated in the multinational HAWK and HARRIER phase 3 trials in 2016. Brolicizumab, a humanised antibody fragment inhibiting VEGF-A, administered 12-weekly, produced comparable anatomical and visual results to eight-weekly aflibercept injections, with similar safety profiles.
Roche and Genetech have also begun phase 3 trials into the intravitreal injection of faricimab, an antibody designed to bind to and deactivate Ang-2 and VEGF-A. It is hoped the bispecific properties of faricimab will result in improved visual outcomes and longer treatment intervals for patients. Phase 2 trials indicated comparable visual outcomes to four-weekly aflibercept injections with faricimab administered every sixteen weeks (Khanani), so signs are positive.
Atrophic AMD gene therapy
Recent success in the treatment of neovascular AMD has advanced gene therapy research for dry AMD. Gene therapy has the potential to intervene earlier than anti-VEGF drugs to reduce progression to more aggressive wet and late stage AMD. However, progress has been slow.
“Dry AMD is a disease that is difficult to treat because we do not fully understand how and why the disease develops in the first place,” said Prof Rakoczy. “I tend to agree with the suggestion that looking at problems in the RPE is a good start, however, until we fully understand some crucial steps in dry AMD development, treatment might be a long time away.”
Several clinical trials have looked at complement protein inhibition as a potential therapeutic approach for dry AMD. To block the inflammation, limit upregulation of angiogenic factors and limit tissue damage, complement inhibitors can be used to suppress the activation of the key elements in the proteolytic cascade. The COMPLETE study, a phase 2 clinical trial with eculizumab (a laboratory-engineered clone of immunoglobulins), however, found it ineffective in the treatment of geographic AMD with no decrease in atrophic lesion growth. The MAHALO study, also phase 2, using lampalizumab, an antibody effective against complement factor D, meanwhile showed a 20% reduction in the size of geographic atrophy lesions. While, phase 3 trials (CHROMA and SPECTRI) with lampalizumab failed to meet the primary clinical endpoint (Holz, Porter).
“The reason it presumably hasn't worked is because that is not the right antibody to target,” explained Associate Professor Andrea Vincent from Retina Specialists and the University of Auckland.
The most positive outcome to date comes from the phase 2 FILLY trial, which reported its results in October last year. A monthly intravitreal injection of APL-2 (a complement C3 inhibitor) resulted in a 29% reduction in atrophy growth when compared to the sham part of the study. Of concern, however, was the treatment group experienced an increased rate of progression to neovascular AMD. A phase 3 trial is now underway (Nebbioso), so we await these results with interest.
These studies all focused on intravitreal injections, but taking a different approach is a combined Ionis-Roche study into IONIS-FB-Lrx, a gene therapy drug administered subcutaneously, which began phase 2 trials this year. This RNA-based drug therapy, also known as antisense technology, works by disrupting the cell’s RNA to turn off a gene. In this case, inhibiting the production of complement factor B (FB) to determine if this will result in less complement activity and decreased FB in the ocular tissue.
Also causing some excitement is the FOCUS study, an open label, dose escalation, multicentre study to assess the safety and biological activity of Gyroscope’s novel therapeutic approach (GT005) in patients with atrophic AMD. Leveraging its gene therapy platform, the company delivered an endogenous anti-inflammatory protein designed to regulate the complement system to a patient’s retina in February this year.
“The idea of this gene therapy is to ‘deactivate’ the complement system, but at a very specific point at the back of the eye, so the patient would otherwise be unaffected by it… (to) slow down the progression of macular degeneration,” explained University of Oxford professor of ophthalmology Robert MacLaren, who carried out the ground-breaking surgery.
At the same time, a research team at the University of Liverpool led by Dr Louise Porter has identified new genes that may be responsible for the development of AMD, which may provide novel targets for disease investigation. Findings from this study identified differentially methylated and expressed genes SKI, GTF2H4 and TNXB that regulate the disease pathways in early and intermediate AMD. Crucially, SKI and GTF2H4 had not been linked to AMD before. Dr Porter’s clinical studies into AMD disease progression show changes in the RPE precede damage to the macular photoreceptors. So, by preventing early epigenetic dysfunction of the RPE, we may be able to arrest disease progression. This latest discovery means therapeutic advancements can focus on altering DNA methylation in the relevant genes and administering treatment via viral vectors directly into the RPE.
The future…
The pathophysiology in AMD is multifaceted, but with research now determining the relevant genetic factors involved, advanced therapies are inevitable. Whilst anti-VEGF medications have been revolutionary for the treatment of neovascular AMD, with gene therapy and antibody inhibition research it will no longer be a case of the ‘ambulance at the bottom of the cliff’ strategy. Therapy will instead focus on early intervention, preventing progression to late stage AMD and current research indicates we may not have long to wait!
References
- Geerlings MJ, de Jong EK, den Hollander AI. The complement system in age-related macular degeneration: A review of rare genetic variants and implications for personalized treatment. Mol Immunol. 2017;84:65-76
- Troutbeck R, Al-Qureshi S, Guymer R. Therapeutic targeting of the complement system in age‐related macular degeneration: a review. Clin Exp Ophthalmol. 05 February 2012, pp 18-26.
- Constable IJ, Blumenkranz MS et al. Gene Therapy for Age-Related Macular Degeneration. Asia Pac J Ophthal: July/August 2016 - Volume 5 - Issue 4 - p 300–303
- Rakoczy EP, Lai CM et al. Gene therapy with recombinant adeno-associated vectors for neovascular age-related macular degeneration: 1 year follow-up of a phase 1 randomised clinical trial. Lancet, 2015 Dec 12:386(10011): 2395-403
- Khanani AM. Simultaneous inhibition of VEGF and Ang-2 with faricimab in neovascular AMD: STAIRWAY phase 2 results. Retina Subspecialty Day, American Academy of Ophthalmology, 2018
- Nebbiosso M, Lambiase A et al. Therapeutic Approaches with Intravitreal Injections in Geographic Atrophy Secondary to Age-Related Macular Degeneration: Current Drugs and Potential Molecules. Int. J. Mol. Sci. 2019, 20(7), 1693
- Dunaief J. Gene Therapy for Macular Degeneration; Scheie Eye Institute, University of Pennsylvania - www.brightfocus.org/macular/article/gene-therapy-macular
- Holz FG, Sadda SR et al. Efficacy and Safety of Lampalizumab for Geographic Atrophy Due to Age-Related Macular Degeneration: Chroma and Spectri Phase 3 Randomized Clinical Trials. JAMA Ophthalmol. 2018 Jun 1;136(6):666-677. doi:10.1001/ jamaophthalmol.2018.1544
- Porter et al. Whole-genome methylation profiling of the retinal pigment epithelium of individuals with age-related macular degeneration reveals differential methylation of the SKI, GTF2H4, and TNXB genes Clin Epigen. (2019) 11:6
Louise Wood is a therapeutically qualified optometrist working at City Eye Specialists in Auckland, New Zealand.