Eye to Eye: quizzing Drs Peter Hadden and Sophie Hill
This month, Drew Jones covers Eye Institute’s Drs Peter Hadden’s and Sophie Hill’s responses to optometrists’ questions about vitrectomy recovery and counselling, anti-VEGF choices and emerging management options for geographic atrophy secondary to AMD. Answers have been abridged.
What does vitrectomy for macula-on retinal detachment involve?
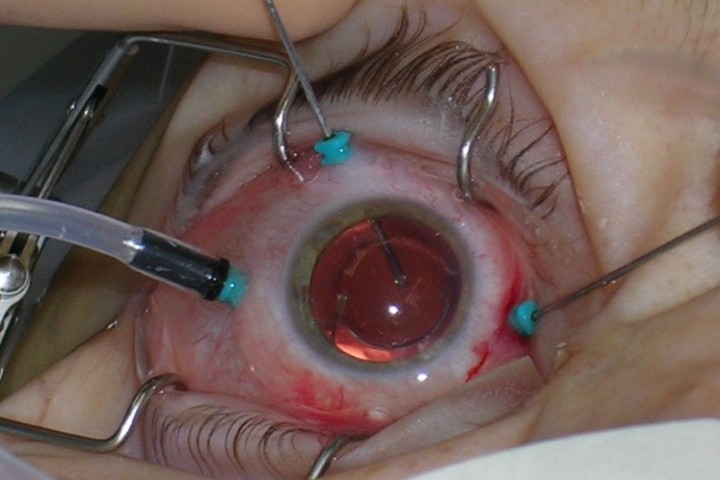
Dr Peter Hadden (PH): A retinal tear is usually caused by vitreous traction with age. Fluid passes through the tear and detaches the retina, like water behind wallpaper. If the macula is still on, that is the point at which you want to catch it because once the macula comes off its blood supply, the vision is never quite the same again.
For a straightforward macula-on detachment, the most common operation is a pars plana vitrectomy: the vitreous is removed, fluid is drained through the retinal hole, then do a fluid-air exchange and leave the eye gas-filled. The tear is sealed with laser, cryotherapy or both. In a simple case, the gas lasts around two weeks and is gradually absorbed into the bloodstream. It is usually about a one-hour day-stay procedure. The patient is awake with light anti-anxiety medication rather than heavy sedation.
Recovery depends partly on the break location. If the break is superior, patients may not need to posture. Otherwise, they may need to lie on the appropriate side for around five days. Vision through gas is like looking underwater. With a two-week gas bubble, patients usually start to see past it after about one week, with the bubble gone at around two weeks. I would not advise driving while gas remains, though desk work is often manageable using the fellow eye. If an optometrist wants to observe surgery, that is fine with patient permission, but macula-on detachments are treated urgently, so they are hard to schedule in advance.

What should we look out for on day one and later review?
PH: On day one, the main issue is pressure. In a gas-filled eye, the retina should be flat and the patient will not see much. A mildly raised pressure, even of 28mmHg, is not usually concerning because it is temporary. A very high pressure may need treatment. A very low pressure generally suggests a wound leak, which usually self-seals. Re-suturing is very uncommon.
At around week three or four, once the gas has gone, the retina is either attached and vision is usually fairly good in a macula-on case, or the retina has redetached and vision will be poor. If a retina redetaches, it tends to do so quickly rather than over a few days. Patients should avoid heavy lifting for four weeks and avoid swimming or opening the eye underwater during that period. A late pressure rise is often a steroid response, so reducing or stopping steroid drops may be needed.
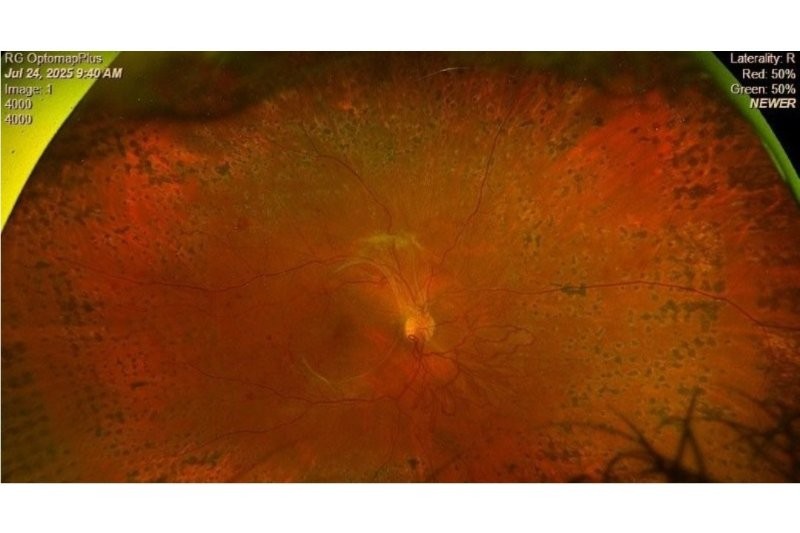
What OCT features and longer-term complications are common after detachment repair?
PH: In a macula-on case, the OCT should usually look normal. One difficulty is that a single-line OCT can be misleading. What looks like oedema may instead be the edge of a lamellar macular hole or another pre-existing feature, so you really need a full set of scans.
An epiretinal membrane is not uncommon after a year or two after a retinal tear or intraocular inflammation, due to haemorrhage and liberated pigment cells settling on the macula over time. In macula-off cases, vision can continue improving for around 18 months, though often not fully back to normal.
Dr Sophie Hill (SH): A single-line OCT is often not enough to make a spot diagnosis. You can miss peripheral disease and other important features without the full scan.
Can needle-phobic patients who need intravitreal injections be sedated?
SH: Usually the first injection is the hardest. Most patients can be managed without sedation if pain is controlled, each step is explained clearly and they have good support. Some clinicians do use short-acting anti-anxiety medication, but that needs appropriate monitoring and backup. In some cases, sedation can actually make the injection harder if the eye becomes less controlled.

How does membrane type influence management in neovascular AMD?
SH: We assess these patients with OCT and usually OCT-A at baseline. Type 1 membranes often have subretinal fluid and can be managed fairly well, with vision often relatively well retained. Type 2 membranes are more associated with fibrosis. Type 3 membranes can respond briskly to treatment but may be more associated with geographic atrophy (GA) over time. Polypoidal choroidal vasculopathy tends to behave more aggressively and can cause significant haemorrhage, so we are more likely to need tighter control and sometimes earlier escalation within funding rules.
Where do anti-VEGF choices sit in New Zealand practice?
SH: Avastin remains first line. Patients usually start with three four-weekly loading injections, then move to treat-and-extend, where possible. Eylea is reserved for a narrower group of patients who have not responded adequately to Avastin, have reacted to it or have certain secondary membranes. Longer-acting options may help reduce treatment burden, but access remains shaped by funding criteria.
Do injections promote GA and are there treatments for GA?
SH: There is debate about whether very aggressive anti-VEGF treatment may contribute to GA progression, which is one reason treat-and-extend is helpful. We do not want to over-treat, but we also do not want to under-treat and leave damaging fluid in the macula. Minimal subretinal fluid may sometimes be tolerated, but only in small amounts.
For GA itself, the discussion is focused on new injectable treatments becoming available overseas, including in Australia, which can modestly slow enlargement of GA but do not stop it. They are most relevant for eyes where the fovea is not yet involved and the goal is to preserve central vision for longer. The benefits are modest and the injections still carry inflammation and vasculitis risks, so uptake depends on careful patient selection and service capacity.
Ed’s note: The trial of Kriya-825, a potential one-time gene therapy for GA, is underway, run by New Zealand Clinical Research.

Laser or injections for diabetic macular oedema and proliferative disease?
PH: For proliferative diabetic retinopathy, panretinal photocoagulation (PRP) remains the mainstay. For diabetic macular oedema, injections are generally preferred, especially for diffuse or centre-involving disease.
SH: A small subset of patients with clearly focal, extrafoveal circinate leakage may benefit from laser. If the oedema is central or diffuse, injections are more appropriate. Needle phobia and patient preference may influence the decision, but foveal-threatening disease is not a laser case. Anti-VEGF can also be used alongside PRP, especially for persistent neovascular activity, though fibrotic eyes can be at risk of ‘crunch’ if injected without adequate PRP.
Can you drive after extensive PRP and can you leave gaps to preserve visual field?
SH: Modern PRP with Pascal tends to be gentler than the old dense burns, so the visual field impact is generally less severe than it used to be. Even so, patients who have had extensive PRP may need Esterman field testing to confirm they still meet driving standards; this should be discussed early.
PRP is a scatter treatment, rather than confluent burns as in retinopexy. Typical spacing is 0.5–0.75 spot diameters, so there is separation between burns and enough collateral retina remains to preserve function. With gentler Pascal PRP, prior treatment can sometimes be hard to see at the slit lamp and may show up better on autofluorescence.
What is the key message for clinicians?
PH: In retinal detachment, timing is everything. If you catch it before the macula is involved, outcomes are much better.
SH: And more broadly, it is about balance. Whether it is injections, laser or new treatments, we are constantly weighing benefit, risk and practicality for each patient.