Implant success for dropless PK
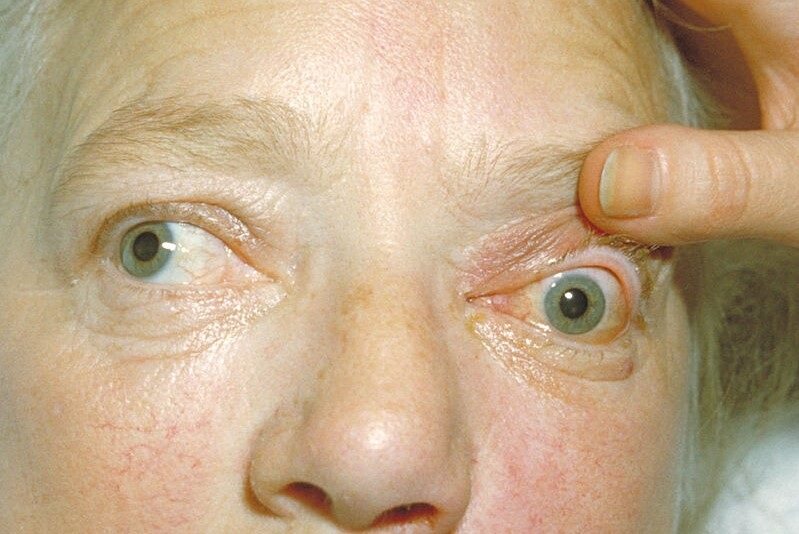
Researchers in France found a subconjunctival dexamethasone implant used instead of steroid eye drops was safe, well tolerated and prevented graft rejection among penetrating keratoplasty (PK) patients.
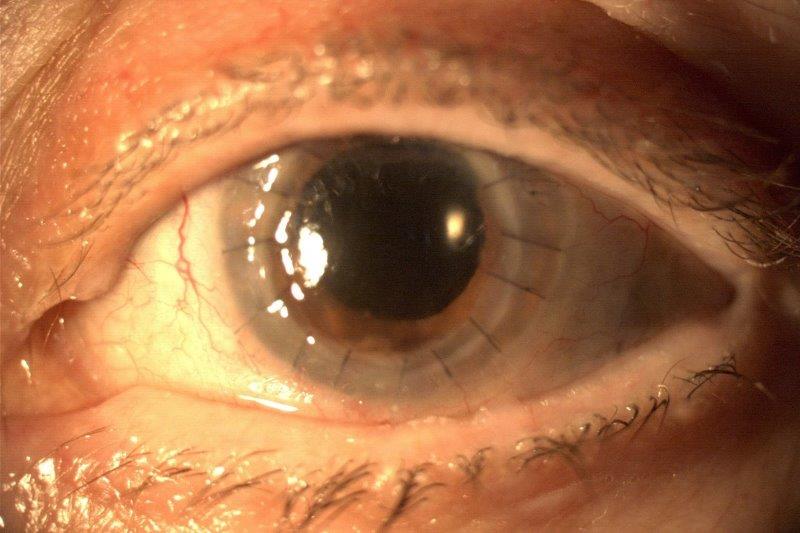
Fourteen low-rejection-risk patients were recruited to the phase 2 pilot, conducted by researchers from University Hospital and Jean Monnet University, Saint-Etienne. After receiving the Ozurdex (Allergan) absorbable implant, loaded with 700μg dexamethasone, no increase in intraocular pressure or other adverse event related to the implant was observed. Following an average resorption time of six weeks, patients were switched to steroid eye drops.
Although the team had previously shown Ozurdex (designed for intravitreal injection to treat macular oedema and noninfectious uveitis) to be well tolerated in rabbits, researchers said this is the first proof of concept that dropless immunosuppression is possible after low-rejection-risk PK.